  
Pickering emulsion biomaterials drug delivery microspheres nanoparticles. Depending on the dispersed and continuous phase, emulsions are primarily categorized into oil-in-water and water-in-oil. Generally, one of the liquid phases is aqueous while the other one is the hydrocarbon or oil phase. This system contains -Cyclodextrin (-CD) and a natural origin selected surfactant, Polyglyceryl-3 diisostearate (PG3D). Emulsions are colloidal systems in which droplets of a liquid phase are dispersed in another liquid phase (Schramm 2014 ). In this article, we give an overview of Pickering emulsions, focusing on some kinds of solid particles commonly serving as emulsifiers, three main types of products from Pickering emulsions, morphology of solid particles and as-prepared materials, as well as applications in different fields. A new oil-in-water (O/W) emulsifying system suitable for use in preparing Pickering emulsions with a large range of oils was developed. Herein, nonadsorbing cellulose nanofibrils (CNF) are shown to induce stabilization of the diluted SLN-stabilized Pickering emulsions via concentration. However, SLN-stabilized emulsions commonly comprise droplets in the micron range, resulting in inevitable instability upon storage. Each image is given in (a and c) the fluorescent channel. Spherical lignin nanoparticles (SLNs) are able to formulate Pickering emulsions with various applications. The three-phase contact angle is the main parameter for evaluating the performance of solid particles. Pickering emulsions are increasingly used in the pharmaceutical and cosmetic fields, especially for topical applications, since these systems require solid particles as emulsifiers instead of surfactants which are known to cause skin irritation. Pickering emulsions can be applied in a wide range of fields, such as biomedicine, food, fine chemical synthesis, cosmetics, and so on, by properly tuning types and properties of solid emulsifiers. CLSM micrographs of hexadecane-in-water emulsion at 10 g/ L OCNF in the water phase: (a and b) without salt and (c and d) with 0.1 M NaCl. Pickering emulsion is mainly formed by the adsorption of solid particles with certain wettability to the oilwater interface, which would form a single-layer or multi-layer film of solid particles. 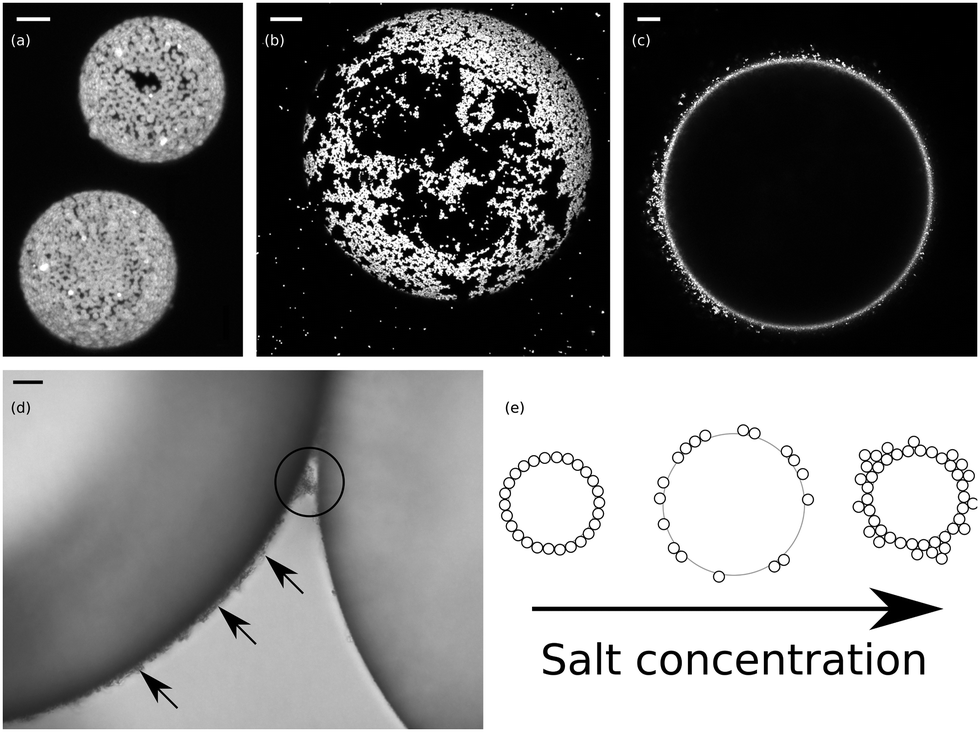
Examples of Pickering emulsions include dry water, a water-air emulsion. Besides, they are more biocompatible when solid particles employed are relatively safe in vivo. experiments showed that CNC should be combined with salt in order to. Substituting solid particles for traditional surfactants, Pickering emulsions are more stable against coalescence and can obtain many useful properties. Pickering emulsion, a kind of emulsion stabilized only by solid particles locating at oil-water interface, has been discovered a century ago, while being extensively studied in recent decades. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
